 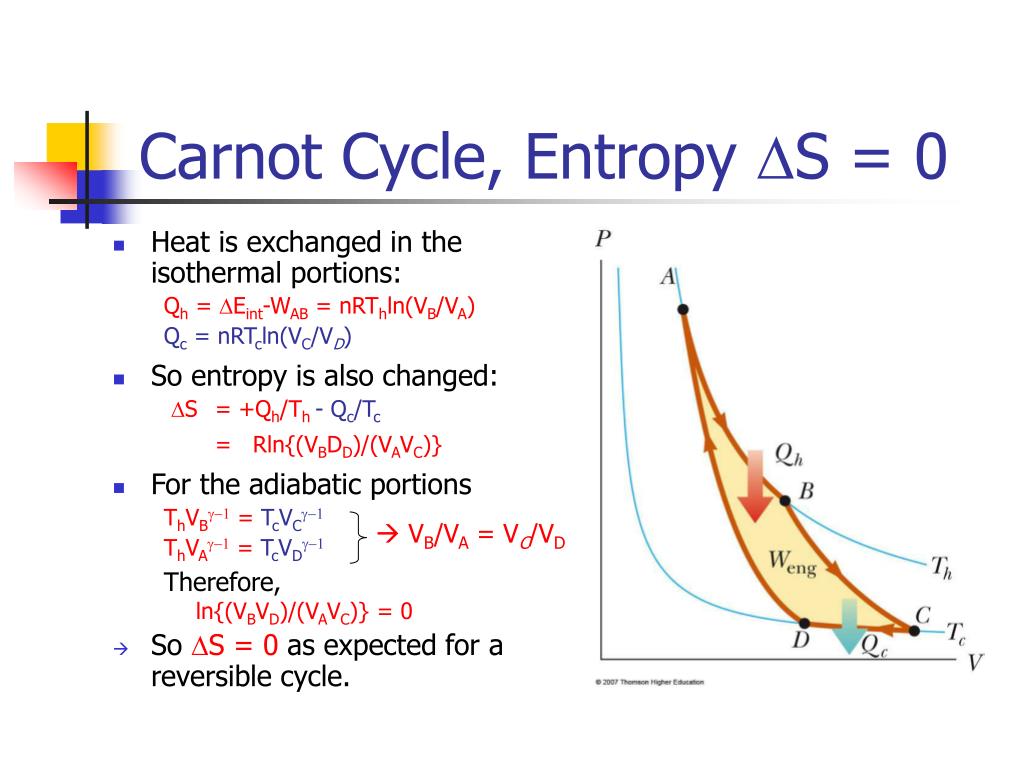
Because this process is very difficult to accomplish in a real device, no heat pump or refrigerator is designed to attempt to approach a Carnot cycle. 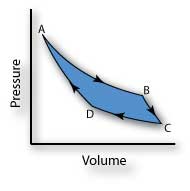
Notice how much the pressure varies during the heat rejection process. The gas is placed in thermal contact with a heat reservoir at a temperature, the heat they furnish is less than the energy used to operate them.From the definition of the Carnot cycle, we have two constant-temperature (isothermal) processes that involve heat transfer and two adiabatic processes in which the temperature changes. The process involved in the Carnot cycle is constant pressure heat rejection, isentropic compression, constant. With an ideal gas as the working substance, the steps of the Carnot cycle, as represented by Figure 4.11, are as follows. Carnot cycle is an ideal cycle in thermodynamics. Finally, because only two reservoirs are involved in its operation, it can be used along with the second law of thermodynamics to define an absolute temperature scale that is truly independent of any substance used for temperature measurement. Yet, it is also very important theoretically, for it plays a major role in the development of another important statement of the second law of thermodynamics. At a practical level, this cycle represents a reversible model for the steam power plant and the refrigerator or heat pump. The Carnot cycle is of special importance for a variety of reasons. If the heat loss from the house is 40kW, the power required to operate the heat pump is a) 1kW b) 2kW c) 3kW d) 4kW Answer: d Clarification: COP(heat pump) Heat loss/Work done T 1 /(T 1 T 2) or, 40/ Work done 273 + 27/. The outside temperature is -3✬ and the indoor temperature is 27✬. An engine operating in this cycle is called a Carnot engine. A carnot heat pump is used to heat a house. In 1824, his studies led him to propose a hypothetical working cycle with the highest possible efficiency between the same two reservoirs, known now as the Carnot cycle. In the early 1820s, Sadi Carnot (1786−1832), a French engineer, became interested in improving the efficiencies of practical heat engines. Demonstrate the equivalence of the Carnot principle and the second law of thermodynamics.Outline the Carnot principle and its implications.Describe the Carnot cycle with the roles of all four processes involved. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
